Proteins
Article curated by Rowena Fletcher-Wood
Proteins are nature's robots, carrying out specific tasks depending on their shape and make-up. Essential in the body for growth, repair, and catalysis, they fight infections, digest food, and perform do all sorts of other functions. They are made up of long chains of amino acids. These amino acids are small organic molecules containing which are linked together using their NH2 and CO2H functional groups. There are only 20 different amino acids that make up all of the proteins in their thousands, and these links fold into and around each other in beautiful, complex ways to give proteins their incredible array of functionality.
Things proteins do
Proteins are central to gene regulation, the process by which genes are switched on and off. When switched on, genes produce a proteins. Different cells and processes require different proteins, and so as a cell specialises necessary genes are switched on and unnecessary ones are turned off.The process is complex and is still not fully understood, with both environmental and lifestyle factors altering which genes get switched on or off (epigenetics/abbr>). Improved knowledge about gene switching could one day allow us to control genes; this could help reduce symptoms of aging and treat disease.


 3
3Believe it or not, proteins are implicated in foetal control of labour; that is, that the foetus may release proteins that promote labour.Researchers have found two proteins in mice that eventually lead to the promotion of labour. These proteins, SRC-1 and 2 (steroid receptor coactivators) produce pulmonary surfactant components, which in turn begins the birth. Researchers found that mice without these proteins waited 2 days longer before labour, a delay that could translate to 1 month in human mothers!

 2
2
Protein technologies


 2
2Proteins also bind to engineered nanomaterials in biological and environmental systems, forming a protein corona. This changes the surface chemistry, reactivity, and fate of the nanomaterials, but it’s hard to know how. If we manage to work this out, it could enable a vast array of pritein nanotechnology, including targeted drug delivery, pollution remediation, and phytotechnology. New predictive models have been created using machine learning and random forest classification[1].


 3
3Protein structure and folding


Proteins also need to be the right shape to work, and they can fold up in milliseconds. But how is not understood any more than what shape they choose. This is a humongous computing problems, as the long chains of amino acids are monstrously long, leading to impossible numbers of alternative folded shapes to identify and test. Although DNA probably contains instructions on how to make and fold proteins, we have not been able to decipher the folding instructions yet.

Interactions such as hydrogen acceptor bonds may play important roles in macromolecular structure and function that have yet to be fully explored and understood. So far, hydrogen acceptor bonds are an unconventional bond type observed in dimers such as borohydride, but not between molecules as a cohesive force.


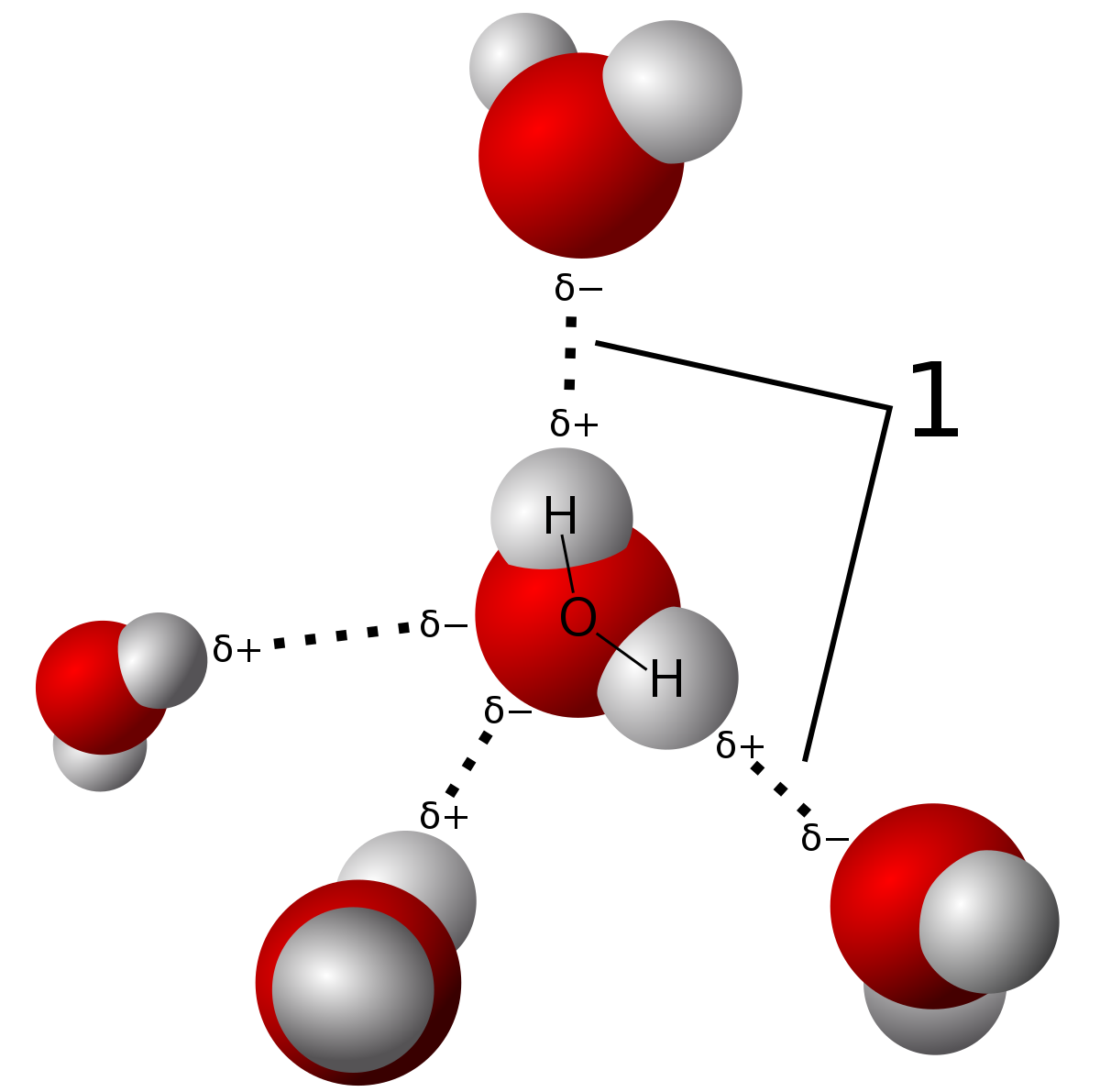


Proteins and evolution


 3
3A recent discovery of a missing gene has left scientists perplexed – why haven't toothed whales been wiped out by disease? The culprits, known as Mx genes, produce proteins that fight off viral infections. In toothed whales, they discovered various mutations that prevented or interfered with protein synthesis. All of the other mammals had normal healthy Mx genes, including the baleen whale, a species that shares a common ancestor with the toothed whales. So, how did these mutations happen and persist? It's possible the common ancestor was exposed to a virus that forced the loss of Mx gene function. This is supported by the findings that viruses today may exploit Mx gene function for individual gains. Scientists are looking to see whether compensatory mutations have evolved to make up for the Mx loss.

 3
3
The evolution of fins to limbs remains a mystery scientists are keen to get to the bottom of. The beginning of an explanation may be found in the discovery of a protein, Gli3, which regulates the development of the fronts and backs of fins (making sure one doesn't outgrow the other!). However, in one set of species evolving limbs, it looks like the backs of fins started growing longer than the front, with more Gli3 present. Whether Gli3 is the only protein involved and whether the results can be applied to other marine life is currently unknown.

 2
2
Diseases
Some diseases have evolved decoy proteins that either (i) slow down the immune system (by disguising themselves so the immune system doesn’t react in time), (ii) distract it into attacking the wrong things (by squirting out decoy proteins in their wake that the immune system attacks preferentially), or (iii) pretending to be harmless host cells by imitating the protein coatings of cells around them. It’s hard to tell what a good immune response against these diseases would be, and as such we're still working on them.


 3
3Interestingly, the SARS-CoV-2 coronavirus uses proteins to mimic host RNA and “silence” distress calls to the immune system. Scientists think that interfering with the protein “nsp14” could be key to defeating coronavirus, giving the body earlier warning and so more time early on to activate antiviral defences. Interfering with “nsp14” might give the body time to fight the virus off before it develops.


 3
3
The Chinese detection of the protein “D-dimer” may be indicative of those at highest risk from coronavirus: the protein has been found at concentrations four times higher than usual in hosts who later die. We're not sure why[2].


 2
2
E. histolytica parasites are covered in proteins known as lectins, which bind to sugars. Inside the body, they bind to sugars on the inside surface of the intestine, where they then do harm. We don't know how they cause harm though: it's certainly not solely down to the protein, which is found in other, harmless microorganisms. Two other proteins are thought to play a role, but we don’t know much about them.
Learn more about Pathogenisis of E. histolytica.

We don't know what causes Alzheimer's, but scientists have noticed plaques and tangles in the brains of sufferers caused by the buildup of tau and beta-amyloid proteins[3].
Beta-amyloid proteins cause plaques, whilst tau proteins cause tangles. Beta-amyloid is usually part of neuronal development, but when there's too much, it stops being soluble and aggregates. When tau proteins pair with too many phosphate groups, clumping occurs and this is now the tangles are made[4].
Healthy brains also contain plaques and tangles, but usually (but not always) nowhere near so many. So are they the result or cause of Alzheimer's? We don't know.
Learn more about Plaques and Tangles in the brain.

![Lateral View of the Brain By BruceBlaus [CC BY 3.0], via Wikimedia Commons](/img/sci/Blausen_0101_Brain_LateralView_512.png)
One recent study found that 75 to 80% of men with dementia with Lewy bodies (proteins in the brain) also experienced REM sleep behaviour disorder – sleepwalking and other movement during sleep. Presentation of the disorder has been strongly correlated with risk of developing dementia with Lewy bodies – the type of dementia seen in Parkinson’s and Alzheimer’s. The correlation is stronger in men than in women, and scientists can’t yet say why it appears to be there[5].


 2
2Porphyria, the “vampire disease” is also all about proteins. A malfunction in the production of haem proteins leading to the build up of porphyrins in the blood, leading to symptoms including receding gums, sensitivity to sunlight and garlic, purple faeces, abdominal cramps, seizures, poor mental health, and muscle and nerve damage. The condition is managed via dietary control. Only 20% of sufferers show a genetic mutation that scientists think may be responsible, however, and the condition in others is attributed to environmental factors like alcohol abuse, although this kind of porphyria also seems to run in families, an observation that cannot currently be explained. It’s also unknown why alcohol, smoking, stress, high iron and oestrogen trigger attacks. Another gene or protein may be involved.


 3
3
.jpg)
Learn more about /smell.


 3
3There are more than 30 proposed mechanisms to explain the action of thalidomide on the body, and in fact, it looks like it does a lot of stuff, causing mutations in DNA and cartilage, interfering with neural networks, generating reactive oxygen species (which accelerate cell ageing in the body), and inhibiting some proteins. Importantly, these proteins are involved in the development of new blood cells, essential for healthy placental formation. The action may be triggered when thalidomide binds to a protein called cereblon. X-rays and electron mapping showed the left-handed version binds easily, but the right-handed version is twisted and easily falls off. To perform these experiments, scientists doped thalidomide with deuterium to stop them from interconverting.


 3
3Prions are pathogens with the same primary amino acid structure as our own proteins. They look like self molecules, but they don’t behave like them, and they trigger normal proteins to fold in abnormal ways. Scientists speculate that vaccines against prions may not be possible, and are still working on how to distinguish them. This would mean changing the paradigm by which we understand pathogen recognition and treatment.


 2
2Proteins are also essential for the proper functioning of our immune system. Cytokines are small proteins that signal between cells. They are produced all over, but in particular are effective immunomodulating agents. In theory, cytokines magnify immune response, but this doesn’t always work out, suggesting there are still things we don’t know about cytokines.Cytokines are different from hormones, but scientists are still unsure how and why. Things they have noticed include that cytokines are produced by a wider variety of cells (pretty much anything with a nucleus), a particular cytokine may be produced by more than one kind of cell, and concentrations vary more than a thousand fold, unusual for hormones or classic proteins. Cytokines may impact just the cell they come from, affect a group of cells, or make changes throughout the entire body – but what governs these differences is unknown. As a possible therapeutic treatment against nerve injury or inflammatory pain, or an agent for vaccination, unweaving the unknowns of cytokines is an important medical challenge.


 3
3
From causing diseases to curing them, to driving evolution, proteins seem to get everywhere. They have amazing potential for medical and technological breakthroughs, and understanding them is an important challenge in science.
This article was written by the Things We Don’t Know editorial team, with contributions from ScienceGremlin, Freya Leask, Lucy Binfield, Ginny Smith, Johanna Blee, Rowena Fletcher-Wood, Joshua Fleming, and Alice Carstairs.
This article was first published on 2021-12-29 and was last updated on 2021-12-29.
References
why don’t all references have links?
[1] Findlay, M. R., Freitas, D. N., Mobed-Miremadi, M., and Wheeler, K. E. (2018). Machine learning provides predictive analysis into silver nanoparticle protein corona formation from physicochemical properties. Environmental Science: Nano, 5(1), 64-71.
[2] Zhang, Litao, et al. D‐dimer levels on admission to predict in‐hospital mortality in patients with Covid‐19. Journal of Thrombosis and Haemostasis 18.6 (2020): 1324-1329. doi: 10.1111/jth.14859.
[3] Lue LF, Brachova L, Civin WH, Rogers J. Inflammation, A beta deposition, and neurofibrillary tangle formation as correlates of Alzheimer’s disease neurodegeneration. J Neuropathol Exp Neurol. 1996 Oct; 55(10):1083–8. doi: 10.1097/00005072-199655100-00008.
[4] Bloom GS. Amyloid-β and tau: the trigger and bullet in Alzheimer disease pathogenesis. JAMA Neurology. 2014 April 71;(4):505-8. doi: 10.1001/jamaneurol.2013.5847.
[5] Murray, Melissa E., et al. (2013) MRI and pathology of REM sleep behavior disorder in dementia with Lewy bodies. Neurology 81.19: 1681-1689.
Blog posts about proteins


Recent proteins News
Get customised news updates on your homepage by subscribing to articles