Smell
Article curated by Ginny Smith
From the heady scent of a rose garden on a summer's day to the rumble in our stomachs that can be triggered by wafts of freshly baked bread as we walk past a bakery, smell plays a huge role in our lives. It is the most complex, the most evocative, and the most mysterious sense. Scientists don't know exactly how we detect different smells, how many of them we can identify, or why they have such a profound effect on our emotions.

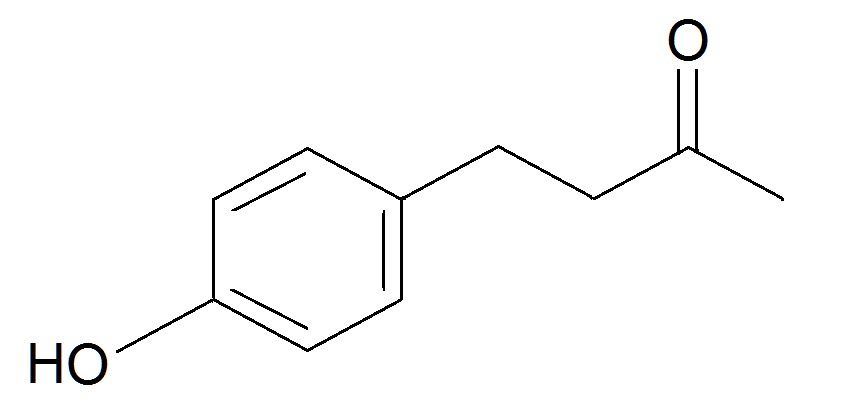
One alternative theory is that of molecular vibration. This suggests that it is not the shape of a molecule but how it vibrates that determines its smell. Convincingly, molecules with very similar shapes but different vibrational frequencies can smell very different[4], and two molecules can smell different if they contain different isotopes - versions of an atom with a different number of neutrons. This changes their vibrational frequency, but not their shape because heavier atoms form more rigid bonds and vibrate at lower frequencies. However, this finding has been criticised because it works in flies, but not humans. Luca Turin, the founder of the theory, thinks this is because of our smell sensitivity. He hypothesises that humans only pick up on the difference in vibrations between normal hydrogen and deuterated bonds when there are 14 or more in a compound, or at least 50% of the hydrogen is substituted by deuterium. Flies, on the other hand, when taught to avoid certain compounds that include deuterium bonds, seemingly avoid all deuterium-containing compounds and, when presented with nitrile compounds, which have a similar vibrational frequency, avoided those too. The theory needs further testing[5].
Learn more about Smell.


 2
2
However others disagree with their finding, claiming the maths is flawed. By simply varying the number of subjects, or the threshold for significance, and re-analysing the original data, the number of distinguishable smells varied dramatically from 4,500 to 100 octillion! The problem arises because smell isn’t easily broken up into categories like, for example, colour, which can be divided by wavelength. Until we work out better how to categorise smell, it seems that the number of discrete scents we can distinguish is impossible to know.
This is made even more complicated because we have different sensitivities to different smells. Some we pick up in tiny concentrations and others merge into the background as noise. It's possible that we have some kind of instinctive recognition criteria, like our preference for human faces - causing us to see faces everywhere (in the foam of a coffee, on the surface of Mars...), or it may be taught, like language. It would not only be fascinating to know more about this, but also incredibly useful, particularly if we can understand it in other animals too: we could replace harmful pesticides in agriculture with smells that disguise edible crops from pests, or make environmentally friendly shark repellants.



At first, scientists were bemused by the COVID-19 coronavirus' effect on sense of smell: whilst many people are asymptomatic, many experience loss or disturbed sense of smell that lasts for months after the illness, before finally returning to normal. And it's not the only illness that can upset our sense of smell. We’re still not quite sure why. However, recent work has found that the coronavirus latches onto two proteins (ACE2 and TMPRSS2) common in the nose[8]. They help transport odour information through mucus, and so it’s thought that when coronavirus binds, it damages the supporting structure around the neuron – the neuron itself is undamaged, and that’s why our sense of smell recovers. Scientists continue to investigate.


 3
3We even have smell and taste receptors in other, rather unusual places - like inside our bodies. Researchers have located sour taste receptors in the spine, sweet taste receptors in the gut and bladder, and bitter taste receptors in the sinuses, airways, brain and sperm. Whilst researchers initially assumed these were accidental, developing in their old haunts long after evolution had improved the human model and reassigned them to the face, amazingly it turned out they were active: smelling and tasting inside our bodies. Although we don’t know why they are there, researchers think they may act as chemical monitors and danger receptors in case we take in something toxic. They might even have originated internally, and only climbed their way up to the mouth and nose more recently.


The world of scent
One thing that hinders our understanding of smell is the complexity of natural odours. Sometimes we can pick up a single molecule, the chemical signature of a distinct smell, like 1,1,6-trimethyl-1,2-dihydronapthalene, which gives the distinctive petrolly flavour to Riesling wines. But while some smells can be recreated easily using a single molecule, others are much more challenging.
One of these tricky smells is that of strawberry. Strawberry flavour is made up of more than 350 odour molecules which built up together so we experience not a mishmash of lots of flavours, but as a single, new flavour. This experience is called synergy, and makes smell science very complicated. In tests on 12 key molecules, participants were asked to smell combinations with just one component missing to see whether they still thought it was strawberry. Six molecules were found to be essential to the strawberry flavour; although it lost dimension if the others were missing, it was still strawberry.



It is thought that 20-30 of the volatile molecules that come off a strawberry create its attractive smell, so what do the others do? Some will be there for ripening or preserving it, and some may attract other animals. Although we don’t know why wild strawberries vary so much in their smells, it is likely to have evolved as the strawberry plant spread to different environments, where its survival depended on appealing to different animals that would feed on the fruit and disperse its seeds. What these animals are and what chemicals they like is still a mystery. However, this does imply that, theoretically, we might be able to breed a strawberry that drives us wild, but that birds turn their beaks up at.


Even when we can determine and re-create the exact chemical makeup of something like a strawberry, it can often smell different to the original. This is because the flesh of the strawberry affects the volatility of the compounds, and so how quickly and strongly they reach our noses. To make a truly realistic smell experience we have to recreate not only the proportions of the molecules present but the patterns in which we detect them. To mix “strawberry flavour” in different foods - bread, for example - we may need to use different amounts of our strawberry molecules than are present in the real thing.


Our sense of smell



Anosmia
It is very rare, but some people are born unable to smell, thanks to genetic mutations which affect the development of the olfactory system It is known as congenital anosmia, and estimates suggest there are 6,000 congenital anosmics in the UK.
We only taste a very small number of basic flavours with our mouths (commonly believed to be five), and this is all people with anosmia (no sense of smell) pick up. In the wild, animals use smell to distinguish safe foods from rotten or poisonous foods before they eat them. Smell is also crucial in locating mates and avoiding predators.
Congenital anosmia can occur alone or it can be a symptom of another genetic condition. Exactly which genes are involved in cases not linked to other conditions isn’t known. One gene which seems to be involved is called Ift88. In mice, knocking out this gene results in the cilia or hairs of their olfactory system being unable to grow, causing anosmia. Researchers were then able to restore the sense of smell in these mice using gene therapy- injecting a virus carrying a working version of the gene into the nose[9]. The mice were then able to use smell to find food. If this finding translates to humans, it might provide hope for anyone whose anosmia is the result of a similar genetic mutation.

 2
2
It’s easy to forget how significant and essential our sense of smell is (78% people in a survey of 49 said if they had to lose a scense it would be smell[10]) - we commonly associate it with only aesthetic things - food tastes and smells, perfumes and nostalgia. But nature seems to think it is more important. Anosmia, whether it is congenital or brought on later in life after infection or head trauma, is a serious social handicap[11]; sufferers often withdraw from social situations like eating out and express feelings of vulnerability and isolation. Men with anosmia typically engage in fewer sexual relationships (3 on average, where the non-anosmic average is 9), and 17% of anosmics score as moderately depressed on the Beck Depression Inventory[12]. Interestingly, most people with disabilities express the same quality of lives as those without after a sufficient time has passed since the onset of the disability, but this doesn’t seem to be the case for loss of smell. This could be due to the links between smell and emotion, or it could simply be that support networks for these sufferers are comparatively poor.



No-one knows exactly how it works, but it is thought that the reorganisation of brain networks may be involved. The cells in the nose are also some of the best in the body at regeneration, so it may be that smell training somehow encourages the regrowth of damaged nerves.

Smell by Instinct
Smell can have a profound effect on behaviour, especially for animals that use it to find food, detect predators and trace mating partners. Many animals use not only scent, but also the vomeronasal organ, which detects pheromones and processes “smell signals” that bypass conscious awareness to produce innate responses. The organ is also known as the “sexual nose”. In mice, this unconscious influence is so powerful that those with their vomeronasal organs damaged never bother to mate. The organ functions seem to go beyond pheromone signalling: snakes even use it to help track prey; however, it is still not very well understood. Wasps, bees and ants use smell as their main form of communication. Forager bees that waggle dance even release information encoded in scent - in the shape of four hydrocarbon molecules - that actively stimulates further bee foraging activity[16]. Whether humans have this organ, and whether it is functional if they do, is a topic of debate. Whilst male hamsters from rival colonies mount each other if sprayed with female hamster pheromones, there is no evidence for it happening with humans! The organ does seem to develop in embryos, but then regresses. Some claim any remnants of it in adults are simply vestigial and non functional. This is supported by the fact that the organ can be found in some adults but not others[17]. Research has also shown that the organ doesn’t contain the required nerve cells to detect input or send signals to the brain.


Even if the VNO is non-functional, as many claim, this doesn't mean there is no way humans can detect pheromones-we may use our main olfactory system. Some studies have suggested substances in sweat can alter the menstrual cycle of women, for example, or increase arousal. How these changes happen remains to be seen.

In animals such as mice and fish, evidence suggests that sexual partners are selected for genetic compatibility (healthy offspring and relationship longevity) using smell - but so far we’re not sure whether the same is true for humans. How genetic identity is encoded in smell molecules is also completely unknown. Human leukocyte antigens share genome space with olfactory receptors, suggesting they are inherited together, so may work together in mate selection, or may have done historically. Smell tests on women sniffing mens’ shirts did produce results consistent with smelling out genetic compatibility. Interestingly, women also outperform men in smell sensitivity tests, which could reflect enhanced partner choosiness as they bear the greater reproductive burden.
If we can detect it, we don’t know how important genetic compatibility is compared with other factors like personality and appearance. Some feel that the answer may be black or white, with mates either wholly driven by instinct or hardly conscious of it, attributing very little significance to it. However it is more likely it would play a role, amongst other factors. Such a mechanism could certainly explain why people often develop chemistry with date partners who do not tick off much on their list of desirable attributes, and why the perfect partner on paper may just not create that 'spark' of romance.


The Smell of Disease

Although there's some evidence that some trained dogs can sniff out the smelly molecules given off by cancers, there have been mixed results on how accurate they are and it's not really practical to think about using dogs on a wide scale to detect the disease.
. It seems unlikely that western states will consider using animals for such detections (though less wealthy states may do so if it proves to be both cheap and effective) it does open up the possibility of developing an electronic nose, or "E-nose", that can detect such cancers. This would potentially be a lot cheaper and much less invasive than the current technique of fine-needle aspiration biopsy[18].

This article was written by the Things We Don’t Know editorial team, with contributions from Ed Trollope, Ginny Smith, Johanna Blee, and Rowena Fletcher-Wood.
This article was first published on 2016-05-12 and was last updated on 2020-10-12.
References
why don’t all references have links?
[1] Axel, R., (2005) Scents and Sensibility: A Molecular Logic of Olfactory Perception (Nobel Lecture) Angewandte Chemie International Edition 44(38):6110-6127. doi: 10.1002/anie.200501726
[2] Buck, L.B., (2005) Unraveling the Sense of Smell (Nobel Lecture) Angewandte Chemie International Edition 44(38):6128-6140. doi: 10.1002/anie.200501120.
[3] Moncrieff, R.W. What is odor? A new theory. Am. Perfumer 54 (1949): 453.
[4] Francoa, M.I. et al., (2011) Molecular vibration-sensing component in Drosophila melanogaster olfaction Proceedings of the National Academy of Sciences 9/108:3797-3802. doi: 10.1073/pnas.101229.
[5] O'Hare, M., (2006) A nose for controversy New Scientist 192(2578):74-75. doi: 10.1016/S0262-4079(06)61148-2.
[6] Bushdid, C., et al., (2014) Humans Can Discriminate More than 1 Trillion Olfactory Stimuli Science 343(6177):1370-1372. doi: 10.1126/science.1249168.
[7] Soudry, Y., et al., (2011) Olfactory system and emotion: Common substrates. European Annals of Otorhinolaryngology, Head and Neck Diseases 128(1):18-23. doi: 10.1016/j.anorl.2010.09.007.
[8] Brann, D.H., et al. Non-neuronal expression of SARS-CoV-2 entry genes in the olfaory system suggests mechanisms underlying COVID-19-associated anosmia. Science Advances (2020): eabc5801. doi: 10.1126/sciadv.abc5801.
[9] McIntyre, J.C., et al., (2012) Gene therapy rescues cilia defects and restores olfactory function in a mammalian ciliopathy model. Nature Medicine 18(9):1423-1428. doi: 10.1038/nm.2860.
[10] Toller, S.V., (1999) Assessing the Impact of Anosmia: Review of a Questionnaire's Findings Chemical Senses, 24(6):705-712. doi: 10.1093/chemse/24.6.705.
[11] Karstensen, H., Tommerup, N., (2012) Isolated and syndromic forms of congenital anosmia Clinical Genetics 81(3):210-215. doi: 10.1111/j.1399-0004.2011.01776.x.
[12] Tennen, H., Affleck, G. and Mendola, R. (1991) Coping with smell and taste disorders. In Getchell, T.V., Doty, R.L., Bartoshuk, L.M. and Snow, J.B. (eds), Smell and Taste in Health and Disease. Raven Press, New York.
[13] Hummel, T., et al., (2009) Effects of olfactory training in patients with olfactory loss. The Laryngoscope 119(3):496-499. doi: 10.1002/lary.20101.
[14] Kollndorfer, K., et al., (2014) Recovery of Olfactory Function Induces Neuroplasticity Effects in Patients with Smell Loss Neural Plasticity 2014:1-7. doi: 10.1155/2014/140419.
[15] Costanzo, R,M., Yagi, S., (2011) Olfactory epithelial transplantation: possible mechanism for restoration of smell Current Opinion in Otolaryngology & Head and Neck Surgery 19(1):54-57. doi: 10.1097/MOO.0b013e328341e242.
[16] Thom, C., et al., (2007) The Scent of the Waggle Dance PLoS Biology 5(9):e228. doi: 10.1371/journal.pbio.0050228.
[17] Meredith, M., (2001) Human Vomeronasal Organ Function: A Critical Review of Best and Worst Cases. Chemical Senses 26(4):433-445. doi: 10.1093/chemse/26.4.433.
[18] Jun, H.H., et al., (2015) Overcoming the Limitations of Fine Needle Aspiration Biopsy: Detection of Lateral Neck Node Metastasis in Papillary Thyroid Carcinoma. Yonsei Medical Journal 56(1):182-188. doi: 10.3349/ymj.2015.56.1.182.
Blog posts about smell




Recent smell News
Get customised news updates on your homepage by subscribing to articles













