Origin of life
Article curated by Joshua Fleming
How did life begin? One of the most basic and yet deeply complex questions ever asked has been met with a multitude of suggestions ranging from religion to science and everything in between, without conclusive evidence ever produced. In this article, we’ll explore some key scientific theories, involving everything from planets to proteins.

4 billion years ago our planet was, unsurprisingly, quite a different place. Volcanoes were forming and erupting, and asteroids were regularly smashing into the surface, gradually shaping the Earth’s surface. A chaotic, volatile and unlikely place for life to begin. But within “just” several hundred million years, begin it did. The earliest chemofossil – a fossil made of the chemicals left after decomposition of the original organism – dates back to around 3.8 billion years ago. So how did it happen? Well, we can look at how it could have happened, but we may never know for sure. Scientists hope that a ‘true’ understanding of the field, as well as progress in artificial biologies, could be gleaned from how life first began. There are two main categories of hypotheses being proposed for the origin of life, replicator first, or metabolism first. Metabolism first hypothesis focuses on simple molecules that could have gradually developed into the complex compounds for life. Conversely, replicator first hypotheses are the idea that naturally occurring molecules could already self replicate and life evolved from these.
To understand how a living organism is built, you first have to know about the blocks needed to build it... and there are three fundamental compounds found in everything alive: nucleic acids, proteins, and lipids. The important nucleic acids for life are deoxyribonucleic acid and ribonucleic acid. They sound complicated, but are usually referred to more familiarly as DNA and RNA! The proteins are effectively the builders, folding into shapes specific to their job within the cell. Finally, lipids are responsible for the general structure of the cell – keeping all of its components inside. Without these molecules cells wouldn’t exist; life wouldn’t exist.

Electric-spark hypothesis
In 1958, the famous researcher Stanley Miller carried out an experiment[1]. Little did he know, it would lead to some of the strongest evidence that supports a replicator first theory. He'd previously theorised that the atmosphere of the Earth billions of years ago contained a combination of the perfect elements and compounds for life to begin, including hydrogen, water, ammonia and methane. These building blocks could join to form amino acids (which are a step between RNA and proteins) and sugars, the precursors to life. All that was needed for this to happen was a spark (from lightning, for example); hence its name, the electric-spark hypothesis. Miller tested this in 1953 with another scientist, Harold Urey, naming it the Miller-Urey experiment. Their resulting samples did contain amino acids (but only five).
When Miller died in 2007, one of his former students (Jeffrey Bader) received some of his old experimental samples from a test that Miller did half a decade after 1953, but for an unknown reason they weren’t analysed at the time. When Bader (among others) began exploring the products, he discovered an astonishing number of amino acids had been produced. The key difference in this experiment to the one in 1953 was that Miller had added one more component to the atmospheric mix: hydrogen sulfide (H2S). Volcanoes naturally release this gas, and so it became conceivable that instead of the elements of life forming in atmospheric clouds with lightning, perhaps life had been sparked in a volcanic cloud.

 2
2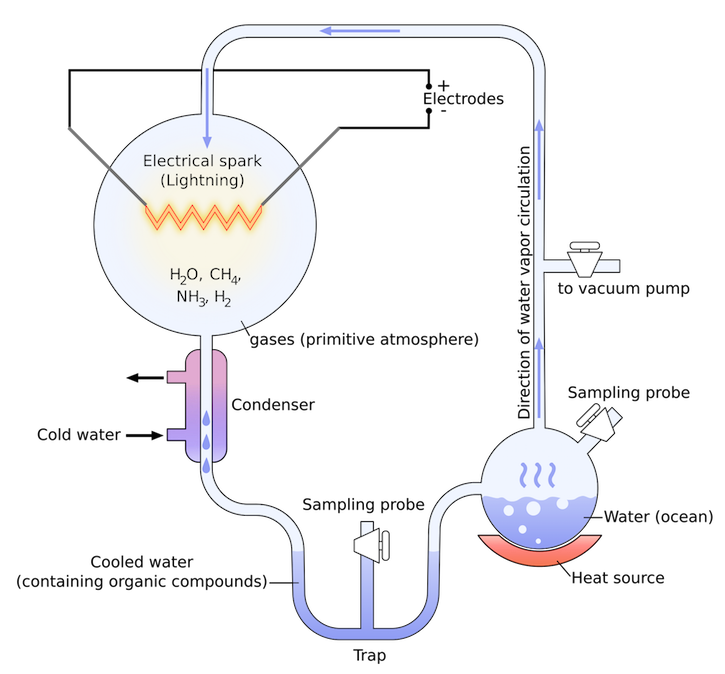
The panspermia theory
Another major proposition is that life originated in outer space. This theory, panspermia, suggests the elements required for life are moving through our universe via asteroids and meteoroids. If these molecules can survive this transportation, then it is feasible that life could be kickstarted with the energy produced from just one collision with our planet.
However, this theory is hard to prove. We don't have many extraterrestrial samples dating from the origins of life and, if we did, we might not be able to tell the difference between rocks acted on by terrestrial life and extraterrestrial life. The only way we could say for sure that life had begun elsewhere would be to find a fossilised record of it trapped within an ancient meteorite. And, even if we did, it would be impossible to rule out life beginning simultaneously and independently here on Earth.
In support of panspermia, researchers have discovered that a combination of compounds not naturally occurring on early Earth could have shaped RNA[4]. Additionally, RNA is unstable in water – which covered (and still covers) the vast majority of the planet. So where are these compounds found? Mars. The research, however, remains inconclusive at present.
Learn more about Location of the Origin of Life.


 3
3
If indeed the building blocks of life came to Earth from outer space, then it should be possible for us to find evidence of these crucial ingredients out there still. Glycine is the smallest of the 20 amino acids that most commonly make up proteins, and is a good example of something to seek. A variety of space probes have been designed to look for it – but one major challenge is that it is very hard to distinguish actual interstellar glycine collected by probes from glycine that contaminated the probe before it left Earth.
In 2009, NASA announced that the Stardust mission had detected glycine in material collected from the tail of a comet called Wild. More recently in 2015, scientists analysing data from the European Philae comet lander reporting finding 16 organic compounds on comet 67P/Churyumov-Gerasimenko, including nitrogen-containing species and four compounds (methyl isocyanate, acetone, propionaldehyde, and acetamide) not previously been reported found on comets. The presence of glycine couldn't be confirmed[5]. Another way of confirming the presence of interstellar glycine is through the analysis of spectral data collected by telescopes, but reported instances of any kind of building block of life, such as amino acids or RNA, have typically been widely disputed.
Learn more about Interstellar glycine.

 3
3Photochirogenesis – the development of chirality in biological molecules – may have arisen due to the interaction of circularly polarised light with racemic molecules generated in meteorites. As such, stars could provide the driving force behind the handedness we now observe in nature, starting in early prebiotic evolution.
Learn more about Photochirogenesis.


RNA world hypothesis
One of the main propositions for the replicator first idea is the RNA world hypothesis. It theorises that RNA was gradually constructed in a mixture of organic molecules that had accumulated on the planet’s surface, known as primordial soup. Critics originally argued that the process just couldn’t occur in the projected conditions on early Earth, but, in 2009, researchers in Manchester produced strong evidence that implied otherwise...[2] They managed to the synthesise the two purines and two pyrimidines that make up RNA using only the materials available on Earth when life started. However, the flaw with this kind of research is not in the chemistry. The flaw is in the logic – that this experimental control by researchers in a modern laboratory could have been available on the early Earth,
[3]. Indeed, the starting molecules, acetylene and formaldehyde, are too complex to be base molecules, and opposition to his research queries where these came from. The team have now gone one step further (back) and discovered chemical pathways to RNA from simpler roots, using only ultraviolet light, hydrogen cyanide (HCN) and the volcanic gas hydrogen sulfide (H2S). They also created proteins and lipids[6]. The research indicated that these reactions still wouldn’t happen all at once, or in one location, but would occur separately and then eventually accumulate. Scientists are looking at how.


Iron-sulfur world hypothesis
On oceanic seafloors all over Earth, there are cracks called hydrothermal vents. Below these is the mantle. Gases including H2S and HCN can travel down through these cracks before being shot back up with torrid water. Nothing evaporates because of the huge pressures at the depths of the fissures. In fact, the gases then dissolve into the heated water around them, creating a biological mixture ideal for the beginnings of life. The iron-sulfur world hypothesis is a metabolism first proposition, and suggests that the first simple molecules were created from this mixture when it flowed over various metals in the rocks surrounding the deep sea vents. Iron and other minerals could have acted as catalysts, fast-forwarding the chemical reactions needed to yield the first simple compounds of life. These could then have been coupled with different metals, creating new aggregates more likely to form yet more complex products.

A linked hypothesis describes the oceans 4 billion years ago as frozen nearly a third of a kilometre thick, due to a less luminous sun. Ultraviolet light would still have been at deadly levels without an ozone layer as protection, but a dense layer of ice may have worked just as well. A cool, stable marine environment might have been ideal for the organic molecules provided by the vents. Additionally, there would be a massively reduced threat from the effects of impact frustration. This is known as the frozen ocean hypothesis.

When did life begin?
It is generally accepted that the Earth formed some 4.54 billion years ago, and that in its earliest days the planet was too inhospitable for life. But when exactly did life emerge? In 2015, UCLA geochemists found evidence that life likely existed on Earth at least 4.1 billion years ago – 300 million years earlier than previous research suggested.
– but is this the final answer? The scientists are sceptical. It is only a possibility, because not all light carbon isotopic signals are biogenic
[7]. We would need to know more about underlying mechanisms to better understand these findings[8][9].


Is the Earth unique?
Because of the huge pressures thought to exist on Ganymede, Jupiter's largest moon, at the ocean floor, scientists previously believed that the layer next to it's rocky core would most likely be ice. This was a problem for the idea that primitive life may have formed on Ganymede at some point in it's history. However, the findings from a NASA study of Ganymede in 2014 imply that the core is next to water – because of the high salinity, denser fluid water would sink toward the core in Ganymede allowing water and rocks to interact.
Ganymede's impressive ocean was first confirmed to exist when NASA's Galileo mission flew by the moon, revealing the water to be hundreds of miles deep. If scientists add salt into the equation they find that the water becomes denser and sinks. This is due to the attraction of water molecules to salt molecules. By using computerised simulations and models, scientists have concluded that a likely composition of Ganymede's ocean is water sandwiched between up to three ice layers, with a salty liquid water layer next to the rocky seafloor.
Studying the oceans of places like Ganymede could reveal more about how life can exist and teach us more about what clues we can look for in the search for alien life.
Learn more about The composition of Ganymede.

 3
3
So, where next?
In the Movile cave in Romania, chemosynthetic bacteria support an entire ecosystem and affect the shape of the cave. Isolated for 5.5 million years and stuffed with toxic gases, scientists think it might reflect the early earth’s environment (but aren’t sure how to be certain). This means the bacteria there may also be similar to some of the earliest life of earth – again, scientists aren’t sure and don’t know how to test this. Not only have huge numbers of new and unusual species been discovered there (many with troglomorphic adaptations for living underground, such as no eyes), but scientists are also keen to investigate how they got into the time capsule. Did they form from 5.5 million years ago, or have they percolated in through the soil more recently?
Learn more about Movile cave.


Alternatively, a proof-of-concept prototype quantum algorithm has produced data that mimics real life biology data pretty accurately. This result, obtained by encoding behaviours linked to self-replication, mutation, and individual interactions, shows that quantum mechanics may be more intimately linked with the origins and nature of life than previously believed. Scientists are still trying to work out how.
Learn more about Quantum origin of life.


 2
2Maybe we’re from a volcano. Maybe we’re from Mars. Maybe we’re from a volcano on Mars… The only thing we know for certain is that this is something we don’t, and may not ever, know.
This article was written by the Things We Don’t Know editorial team, with contributions from Ed Trollope, Jon Cheyne, Freya Leask, Cait Percy, Grace Mason-Jarrett, Rowena Fletcher-Wood, and Joshua Fleming.
This article was first published on 2015-09-04 and was last updated on 2021-06-14.
References
why don’t all references have links?
[1] Miller, Stanley L. A production of amino acids under possible primitive earth conditions. Science 117.3046 (1953): 528-529. doi:10.1126/science.117.3046.528.
[2] Powner, Matthew W., Béatrice Gerland, and John D. Sutherland. Synthesis of activated pyrimidine ribonucleotides in prebiotically plausible conditions. Nature 459.7244 (2009): 239-242. doi:10.1038/nature08013.
[3] Van Noorden, Richard. RNA world easier to make. (2009). doi:10.1038/news.2009.471.
[4] Benner, Steven A., Hyo-Joong Kim, and Matthew A. Carrigan. Asphalt, water, and the prebiotic synthesis of ribose, ribonucleosides, and RNA. Accounts of chemical research 45.12 (2012): 2025-2034. doi:10.1021/ar200332w.
[5] Goesmann, Fred, et al. Organic compounds on comet 67P/Churyumov-Gerasimenko revealed by COSAC mass spectrometry. Science 349.6247 (2015). doi: 10.1126/science.aab0689.
[6] Patel, Bhavesh H., et al. Common origins of RNA, protein and lipid precursors in a cyanosulfidic protometabolism. Nature chemistry 7.4 (2015): 301-307. doi:10.1038/nchem.2202.
[7] Bell, Elizabeth A., et al. Potentially biogenic carbon preserved in a 4.1 billion-year-old zircon. Proceedings of the National Academy of Sciences 112.47 (2015): 14518-14521. doi: 10.1073/pnas.1517557112.
[8] McCollom, Thomas M. Laboratory simulations of abiotic hydrocarbon formation in Earth’s deep subsurface. Reviews in Mineralogy and Geochemistry 75.1 (2013): 467-494. doi: 10.2138/rmg.2013.75.15.
[9] Shilobreeva, S., et al. Insights into C and H storage in the altered oceanic crust: Results from ODP/IODP Hole 1256D. Geochimica et Cosmochimica Acta 75.9 (2011): 2237-2255. doi: 10.1016/j.gca.2010.11.027.
Recent origin of life News
Get customised news updates on your homepage by subscribing to articles














